
Advancing precision drug through innovative chemistry and advanced molecular technologies.

Integrated CDMO services spanning discovery, process development, custom synthesis and commercial manufacturing.

Delivering reliable CDMO solutions for innovative therapeutics worldwide.
Organic synthesis, custom synthesis, process development, and scale-up support for advanced chemistry projects.
Leveraging advanced synthesis technologies and analytical expertise to enable efficient, scalable, and sustainable development of complex molecules.
CHEMOS is a synthesis-driven CDMO focused on organic synthesis, custom synthesis, route development, process development, scale-up support, and analytical support. We work across advanced drug-delivery materials, bioconjugation building blocks, radiopharmaceutical chelators, targeted protein degradation intermediates, and specialty organic compounds. Supported by an experienced R&D team and production base, we help customers evaluate routes, prepare project-specific materials, and plan practical scale-up work.
Explore the latest news and insights from Chemos, including industry trends, event updates and customer case studies.
Chongqing Xingcan Pharmaceutical Technology Co., Ltd. (CHEMOS) will participate in CPHI China 2026, held on 16–18 June 2026 at SNIEC, Shanghai (Booth No.: W9C42). The company will showcase its small molecule CDMO services, advanced drug delivery materials (lipids, PEG derivatives, linkers, functional excipients, etc.), and process development & manufacturing capabilities. CHEMOS warmly welcomes global partners, clients, and industry professionals to discuss custom synthesis, CDMO cooperation, material sourcing, scale-up manufacturing, and long-term strategic partnerships.
Read MoreTrifluoromethanesulfonic anhydride (Tf2O) is a highly efficient electrophilic activator in modern pharmaceutical synthesis, enabling precise introduction of fluorine atoms or trifluoromethyl groups into molecular scaffolds. This enhances metabolic stability, lipophilicity, and biological activity—key parameters in drug discovery. Its mechanism involves converting alcohols into reactive triflates, facilitating nucleophilic substitutions and cross-couplings. As a professional CDMO, we offer custom synthesis of Tf2O at ≥99% purity, scalable from gram to kilogram and beyond, with stringent quality control and fast delivery. Our services help pharmaceutical companies streamline fluorination steps, shorten synthetic routes, reduce costs, and accelerate the transition from lab to commercialization for novel fluorinated APIs.
Read More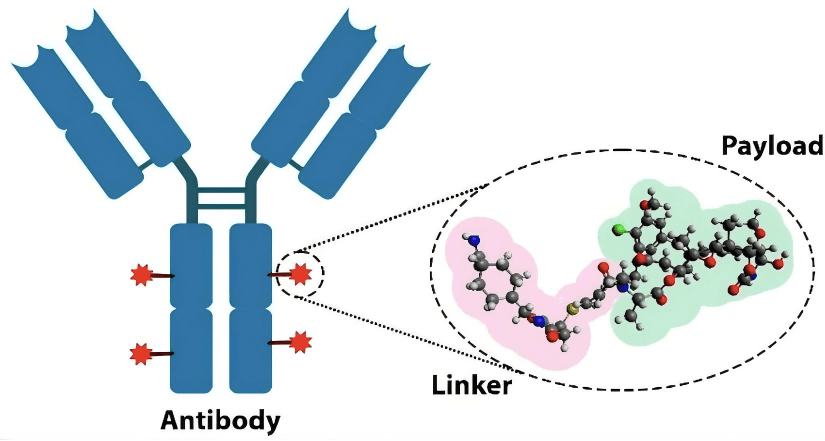
Antibody-drug conjugates (ADCs) enable targeted cancer therapy by linking antibodies to cytotoxic drugs, realizing Paul Ehrlich’s “magic bullet.” A recent review highlights linkers as key to stability and efficacy: cleavable linkers (e.g., enzyme-sensitive Val-Cit) enable precise tumor release, while non-cleavable linkers offer better plasma stability. Core strategies include site-specific conjugation, release triggers, and hydrophilic spacers to prevent aggregation. Advances feature dual-stimuli triggers, traceless release, branched structures, and AI tools like Linker-GPT. With 17 ADCs approved, challenges remain in immunogenicity, manufacturing, and resistance; future efforts focus on computational modeling and nanotechnology to expand applications beyond oncology.
Whether you're in early discovery or preparing for scale-up, CHEMOS delivers efficient, reliable, end-to-end CDMO support powered by advanced synthesis technologies.